Published: May 2026
Read time: 4 min
Published: May 2026
Read time: 4 min
Olivier Duhaime has run hundreds of kilometres across North America. He has completed ultramarathons and four Ironman triathlons. Born in Montreal and raised in Hull, Quebec, Olivier is proud of his home province, while also deeply connected to the National Capital Region — a connection he celebrated by running 400 kilometres unassisted from Quebéc City’s Parliament Building to Ottawa’s Parliament Hill for Canada’s 150th anniversary.
Starting with his first marathon at just 18 years old, Olivier has always pushed his body to the limits. But in the fall of 2021, something started to slow him down. A dull, persistent pain was building in his back. At first, Olivier tried to ignore it. Like many athletes, he was used to pushing through discomfort. But there were also some strange symptoms. For instance, just a sip of beer left him needing to lie down.
As the months passed, the pain worsened. “I’ve run Ironmans and ultramarathons. I know my body,” says Olivier. “And this just didn’t feel right.”
Late one night in February 2022, the pain became unbearable. Olivier decided to drive himself to the Gatineau Hospital.
What he learned next changed his life forever.
“I just never imagined it could happen to me.”
Doctors ordered a CT scan to better understand what was causing Olivier’s pain, and the results were shocking.
Scans revealed three tumours — two in his back and one near his chest. At just 32 years old, the lifelong endurance athlete and father of an eight-month-old daughter was faced with something he had never imagined. Cancer.
In that moment, everything Olivier thought he knew about his health, his future, and his young family shifted. “There’s no history of cancer in my family,” he says. “It was never something I really thought of or knew much about. I just never imagined it could happen to me.”
As he drove home from the hospital the next day, the reality of the diagnosis began to sink in. Along the way, he passed a family skating outdoors with their young daughter. In that moment, Olivier couldn’t help but wonder if he would be alive to teach his own daughter how to skate one day.

Facing a complex diagnosis
In the days that followed, Olivier underwent a series of tests and biopsies to help doctors better assess the disease and determine the best course of treatment.
The results confirmed the diagnosis: Stage 4 Hodgkin lymphoma.
Olivier began chemotherapy treatment at the Gatineau Hospital Cancer Centre, and while it was physically and emotionally demanding, scans showed promising results. Olivier felt a glimmer of hope that he was beating cancer, but the relief was short-lived.
By the fall, scans revealed the cancer had returned — stronger and more aggressive than before. Olivier’s care team recognized his condition required highly specialized treatment and referred him to The Ottawa Hospital Cancer Centre, the region’s leader in advanced cancer care and research.
Specialized care at The Ottawa Hospital
At The Ottawa Hospital, Olivier was greeted by a multidisciplinary team of hematologists, nurses, and specialists who quickly reviewed his scans, biopsies, and treatment history. They designed a personalized plan tailored to treat his aggressive lymphoma through intensive chemotherapy and a bone marrow transplant performed by the highly specialized transplant team.
“She told me, ‘Maybe at other hospitals your cancer would be considered complicated. But for us, it’s something we treat every day.’ That’s when I felt hope again.”
— Olivier Duhaime
For Olivier, this expertise was deeply reassuring.
“I’ll never forget the nurse who first met me at The Ottawa Hospital,” says Olivier. By then, he had made a will, expecting the worst. “She told me, ‘Maybe at other hospitals your cancer would be considered complicated. But for us, it’s something we treat every day.’ That’s when I felt hope again.”

Olivier began an intensive round of chemotherapy, eight hours per day for a week straight, leaving him exhausted, weak, and completely dependent on his care team. This wasn’t the routine chemotherapy he had received before.
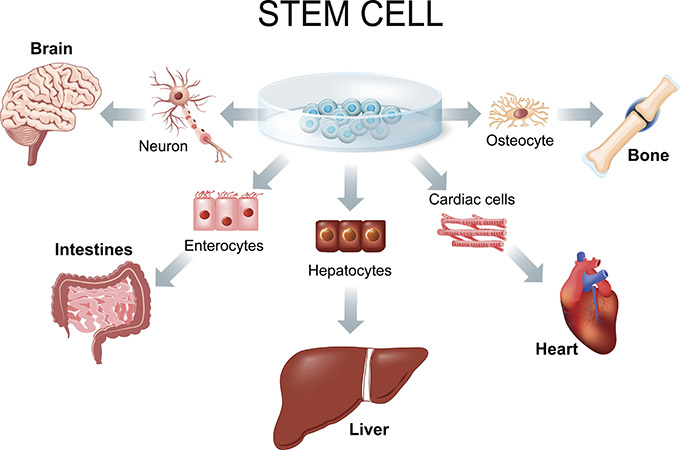
“The high-dose chemotherapy we give before transplant is very powerful and is meant to target the lymphoma,” explains Dr. Ram Vasudevan, one of the physicians responsible for Olivier’s care. “But it also wipes out the bone marrow. We reintroduce the patient’s own stem cells, which act like seeds. They find their way back to the bone marrow and begin rebuilding a new, healthy blood and immune system. But the period between chemotherapy and the bone marrow regrowing is very critical — by complications of not having a functioning bone marrow, but also by the complications and side effects of high-dose chemotherapy at the same time.”
Although this treatment was difficult, Olivier had something unexpected keeping him focused. From his hospital bed, he knew runners were gathering in downtown Ottawa for Tamarack Ottawa Race Weekend, one of Canada’s largest running events, and one he had participated in before. As he watched runners cross the finish line on television, he promised himself that one day he would be back in the race.
A second chance at life
On May 18, 2023, Olivier reached what transplant teams call “Day 0” — the day he would receive his stem cells for his bone marrow transplant. Doctors infused Olivier’s own healthy stem cells into his bloodstream, where they travelled to his bones and began the extraordinary process of rebuilding his bone marrow and immune system.
All about exercise and mental health
“Day 0 is the start of a brand-new life.”
— Olivier Duhaime
“For transplant patients like me, it marks an important day. You’re basically a newborn again,” says Olivier. “Day 0 is the start of a brand-new life.”
Now, Olivier celebrates two birthdays each year: the day he was born in March and May 18, the day he received his transplant and a second chance at life.
Recovery was slow and uncertain. His body had to rebuild its strength and immune system from scratch. But step by step — literally — Olivier began to reclaim his life.
And eventually, he started to run again.
Giving back through Race Weekend
Running has aways been a big part of Olivier’s life, so returning to it, even if it was just short distances at first, was incredibly meaningful.
And just like he had promised himself, Olivier will run the marathon in this year’s Tamarack Ottawa Race Weekend. This time, he’s running for a new reason — to give back to the hospital that saved him Olivier is encouraging friends, family, and the community to fundraise in support of The Ottawa Hospital and the lifesaving care it provides to patients facing cancer, just like him.
“I’m alive because of this hospital and the people who support it.”
— Olivier Duhaime
Gratitude and hope
This spring, Olivier and his partner Justine, are preparing to welcome their second child — a moment Olivier once thought he might not get to experience.
He credits the expertise, compassion, and dedication of his care team at The Ottawa Hospital, especially the Transplant and Cellular Therapy Program, and the generosity of donors, for giving him his life back.
“I’m alive because of this hospital and the people who support it,” he says. “Every step I take now, every race I run, reminds me of the second chance I’ve been given.”